|
The component resolved diagnosis (CRD) marked the beginning of molecular allergology era.
The immunofluorescence two-step indirect method and biochip
technology are used for the component resolved diagnosis.
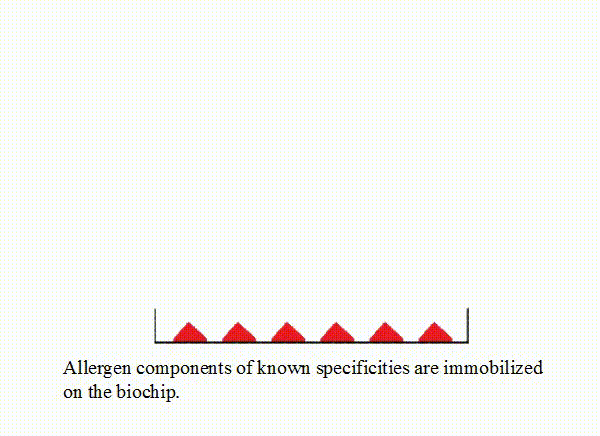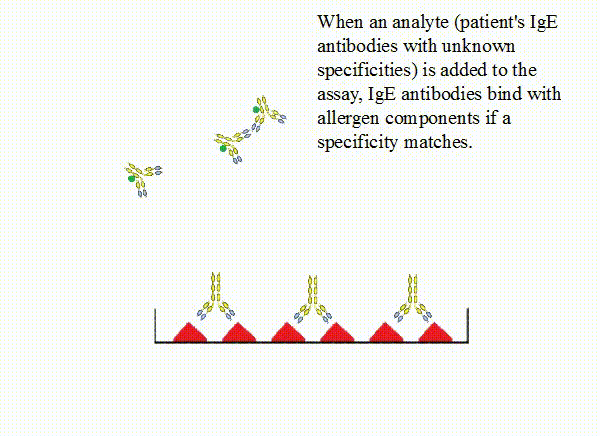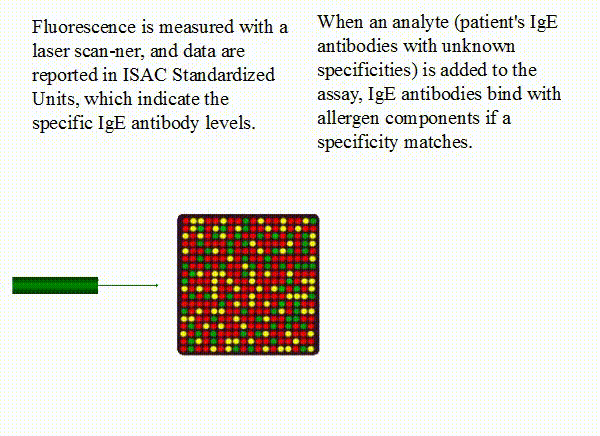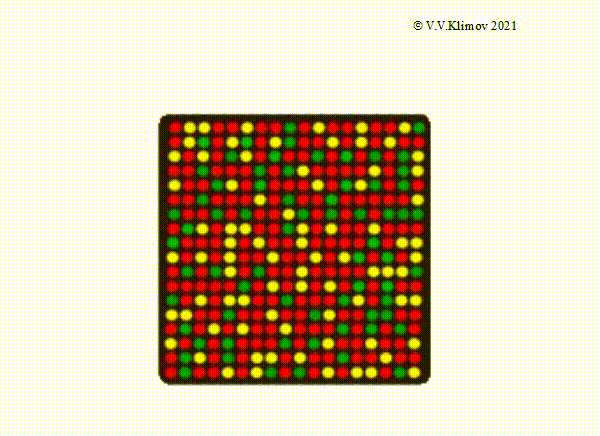
Allergen components of known specificities are immobilized on the biochip.
When an analyte (patient's IgE antibodies with unknown specificities) is
added to the assay, IgE antibodies bind with allergen components if a specificity matches. In contrast, other
IgE antibodies will be removed upon a wash step of the assay. After that, fluorophore-labeled
anti-IgE antibodies react with allergen+IgE forming larger complexes allergen+
IgE+anti-IgE+fluorophore. Fluorescence is measured with a laser scanner, and data are reported
in ISAC Standardized Units, which indicate the specific IgE antibody levels.
©V.V.Klimov
|